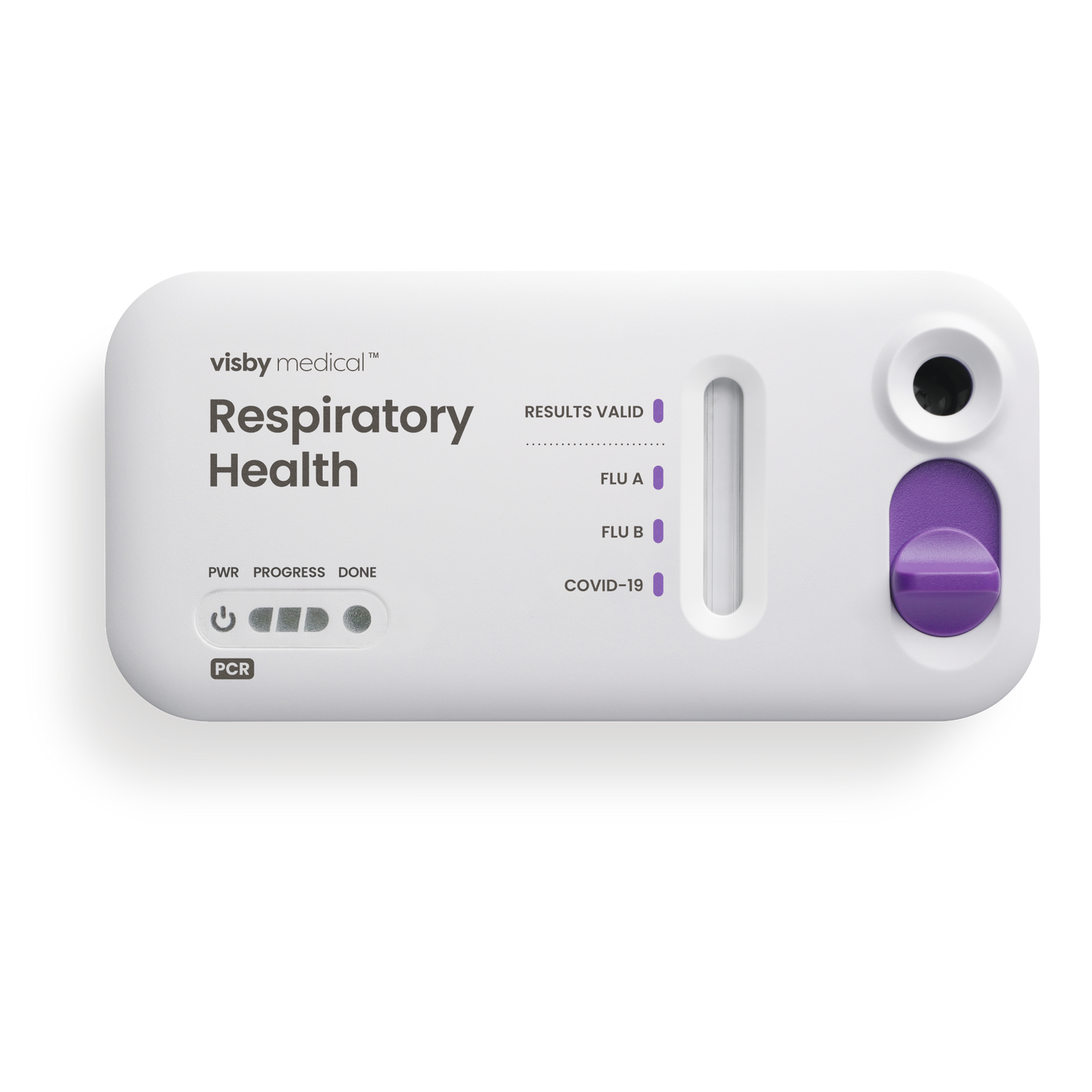
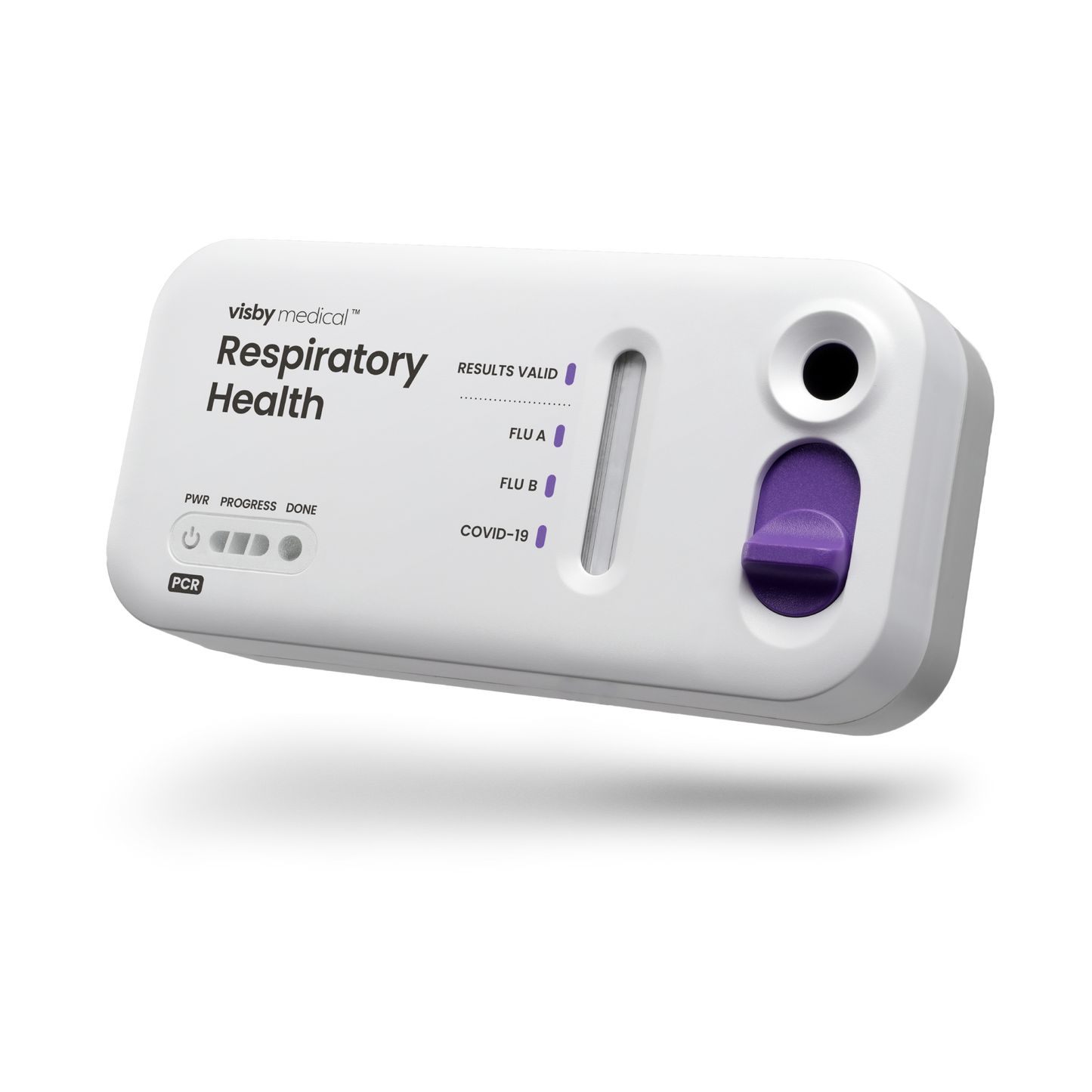


Instrument-free PCR for respiratory health
- Easy to use
One swab, three targets: COVID-19, influenza A and influenza B - Affordable
No maintenance or service contracts - Scalable
Easy to run multiple test devices at the same time
Three critical targets
COVID-19 and flu are highly contagious respiratory illnesses. The Visby Medical Respiratory Health Test can differentiate between the causative viruses and give answers to help guide providers toward the most effective treatments.
-

Flu A
SourceAccording to the CDC, molecular assays, including RT-PCR, are recommended for testing respiratory tract specimens from hospitalized patients because of their high sensitivity and high specificity.
-

Flu B
SourceThe Infectious Diseases Society of America (IDSA) recommends use of rapid influenza molecular assays over rapid influenza diagnostic tests (RIDTs) for detection of influenza viruses in respiratory specimens of outpatients.
-

COVID-19
SourceThe current gold standard is to perform reverse-transcription polymerase chain reaction (PCR) on nasopharyngeal samples. Best-in-class assays demonstrate a limit of detection (LoD) of approximately 100 copies of viral RNA per milliliter of transport media.

Scale as surges happen
The instrument free Visby Medical Respiratory Health Test allows testing for COVID-19 and flu where it’s most effective, at the point of care.
How to use the Visby Medical Respiratory Health Test
Less than 15 seconds of hands-on time. Results in under 30 minutes.
Product Accuracy
| Limit of Detection (LOD) | Influenza A | Influenza B | SARS-CoV-2 |
| Nasopharyngeal Swab | Influenza A/HINI 2009, Brisbane/02/18 106 copies/swab Influenza A/H3N2, Kansas/14/2017 125 copies/swab |
Influenza B/Washington/02/19 728 copies/swab Influenza B/Oklahoma/10/2018 778 copies/swab |
SARS-CoV-2 (USA-WA1/2020) 100 copies/swab |
| Positive Percent Agreement (PPA) | 97.9% | 96.9%* | 97.4% |
| Negative Percent Agreement (NPA) | 99.3% | 100%* | 98.8% |
* Data is a combination of prospective fresh specimens (NP and AN), banked specimens (NP), and surrogate specimens (NP).
Powerful and easy to use, anywhere
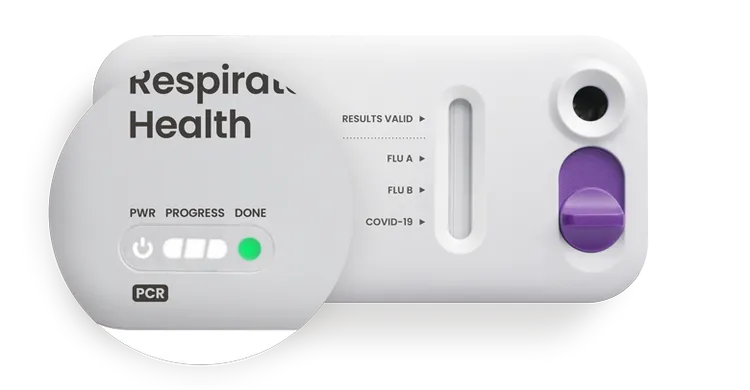
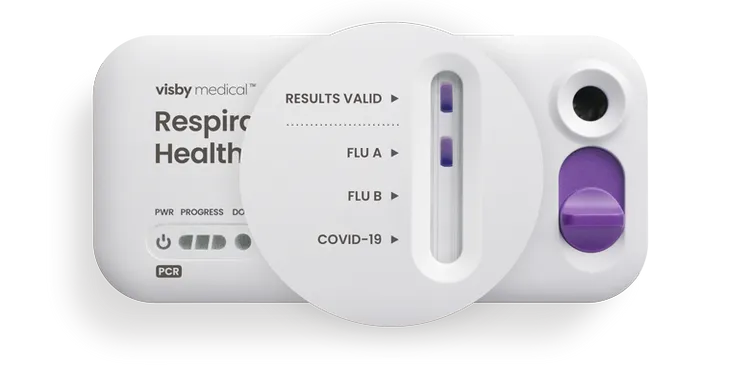
LED Status Lights
Power, progress and ready lights indicate the status of the test.
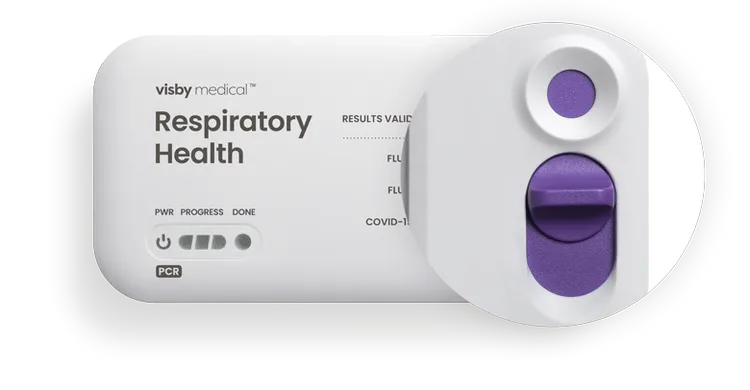
Sample Port
Add the sample and slide the purple switch to run the test.
Test Control
A clear, identifying spot that indicates test results are valid.
Downloads
Visby Medical developed PCR easy enough to be used by almost anyone, in any CLIA-waived setting. Below are resources to help run a successful test.
Instructions for Use
Studies
-

Visby Medical™ Sexual Health Test Results in More Appropriate Antibiotic Treatment
Read full study05/30/2024 • Press Release
Point-of-Care test significantly shortens time from ED arrival to test results, treatment and discharge – significant improvements are seen in the use of antibiotics for the treatment of chlamydia and gonococcal infections in women. Nationwide increases in sexually transmitted diseases and antibiotic resistance create the need for a paradigm shift from traditional lab-based molecular testing.






