
Test, talk and treat - in the same visit.
For self-collected vaginal swabs
- Accurate
Delivers ~97% accuracy. See our instructions for use. - Fast
Results in under 30 minutes, while the patient is present. - CLIA-Waived
Easy to use at the point-of-care; no extra instrument or maintenance needed. - Antibiotic stewardship
Avoid unnecessary antibiotics.
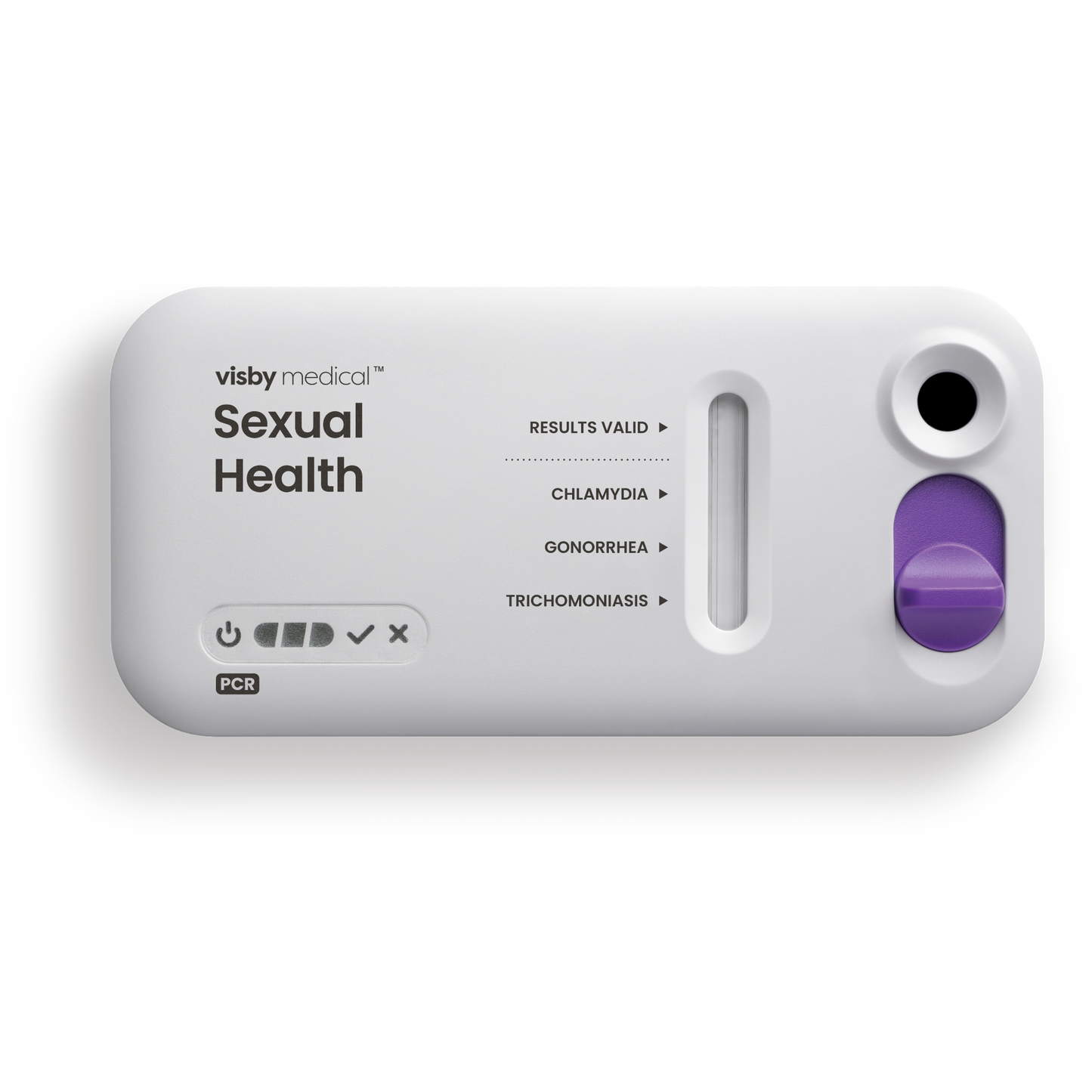
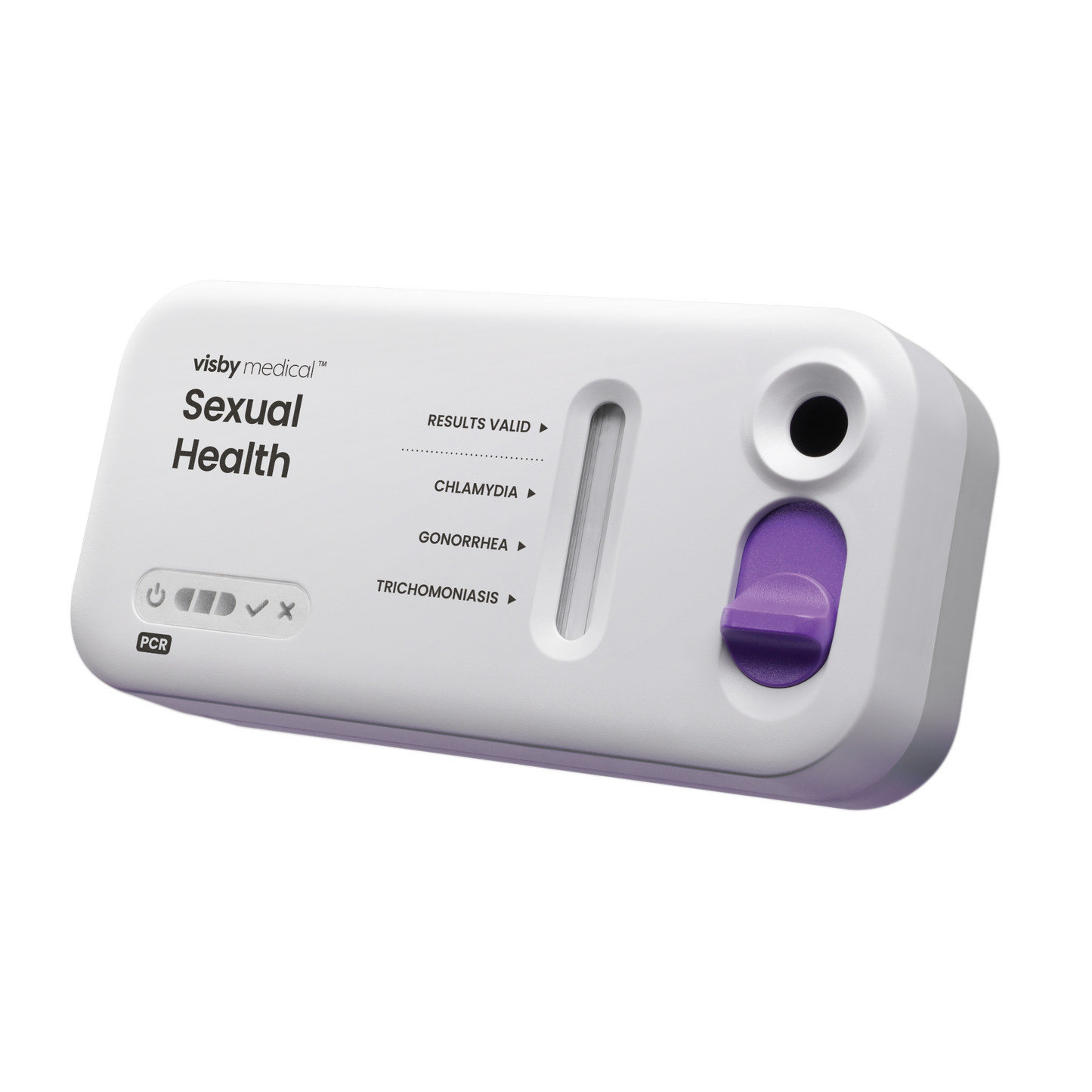
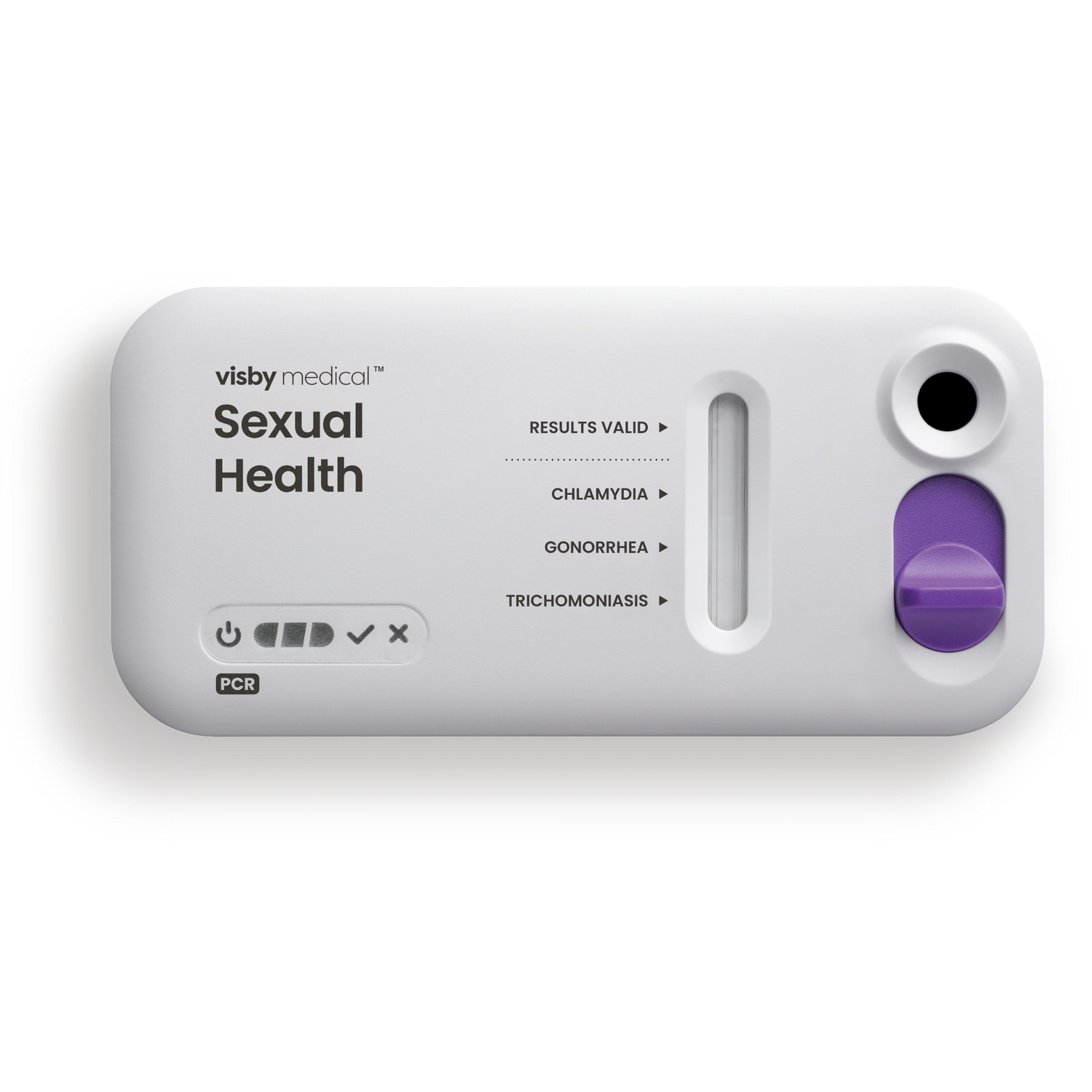
Visby Medical PCR
Game-changing: The first true PCR point-of-care device for STI that’s all-in-one and requires no separate instrument or reader. Results available during the patient’s visit help to eliminate the guesswork of empirical medicine and patient callbacks associated with lab send outs. With the confidence of a PCR result, clinicians can prescribe the correct treatment and engage in a clear, guiding discussion with the patient.
Three critical targets
Our sexual health test targets three pathogens that, if left untreated, can cause permanent damage to a woman’s reproductive system, including infertility, or potentially causing fatal ectopic pregnancy (pregnancy that occurs outside the womb). Fast PCR can help with early detection of these serious infections.
-

Chlamydia
The most common reportable bacterial STI in the US2. Often without symptoms but is associated with significant healthcare costs, affects people of all ages, and is most common in young women.
2. National overview – sexually transmitted disease surveillance, 2019. Centers for Disease Control and Prevention (CDC) website
-

Gonorrhea
The second most common reportable condition in the US, this bacterial STI3, if untreated, may cause infertility. Babies of infected mothers can be infected during childbirth.
3. National overview – sexually transmitted disease surveillance, 2019. Centers for Disease Control and Prevention (CDC) website
-

Trichomoniasis
Caused by a protozoan parasite, Trichomoniasis is the third most common STI in the US4, yet most do not realize they are infected. Trichomoniasis increases the risk of contracting other STIs, including 2-3 times greater risk of HIV acquisition5.
4. Microb Cell. 2016;3:404-418
5. J Infect Dis. 2007;195:698-702
How to use the Visby Medical Sexual Health Test
Less than 15 seconds of hands-on time. Results in under 30 minutes.
Product Performance
| Positive Percent Agreement (PPA) | Negative Percent Agreement (NPA) | |
| CT | 97.4% | 97.8% |
| NG | 97.8% | 99.1% |
| TV | 99.3% | 96.7% |
Powerful and easy to use, anywhere

LED Status Lights
Indication lights inform testing operation status.
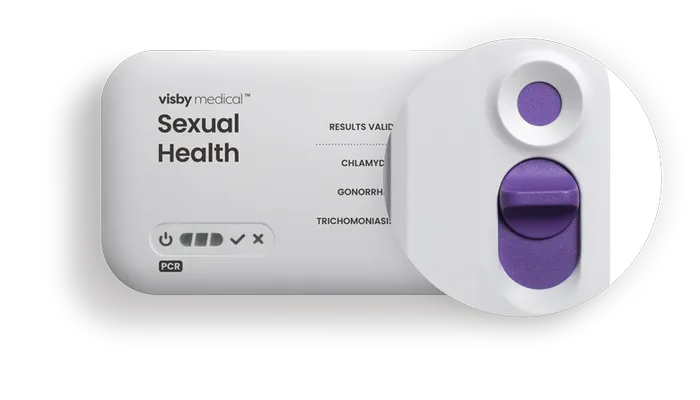
Sample collection port
An easy-to-slide switch secures the patient sample in the collection port.

Test control
A clear identifying spot that indicates test results are valid.
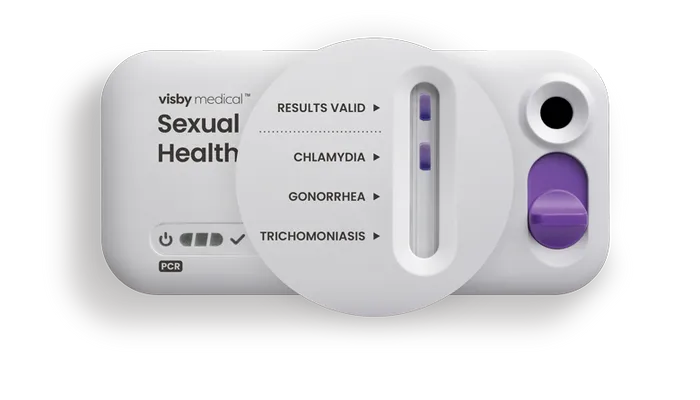
Quick, accurate results
With ~97% accuracy*, a spot next to the pathogen provides assurance that the patient sample is positive.

Downloads
Visby Medical developed PCR easy enough to be used by almost anyone, in any CLIA-waived setting. Below are resources to help run a successful test.
Studies
-

Visby Medical™ Sexual Health Test Results in More Appropriate Antibiotic Treatment
Read full study05/30/2024 • Press Release
Point-of-Care test significantly shortens time from ED arrival to test results, treatment and discharge – significant improvements are seen in the use of antibiotics for the treatment of chlamydia and gonococcal infections in women. Nationwide increases in sexually transmitted diseases and antibiotic resistance create the need for a paradigm shift from traditional lab-based molecular testing.






